The Drug Enforcement Administration (DEA) is now calling for the production of even more THC, psilocybin and DMT for research purposes than it initially proposed for 2024—raising its quotas for those drugs while maintaining already high production goals for marijuana and other psychedelics.
In a notice set to be published in the Federal Register on Wednesday, DEA said it received comments from registered manufacturers requesting increases to previously proposed 2024 quotas for the Schedule I substances in order to “meet medical and scientific needs,” and it agreed to do so in the new final order.
Accordingly, the agency nearly doubled the quotas for delta-9 THC and all other tetrahydrocannabinol, increasing them to 1,523,040 grams and 1,166,130 grams, respectively.
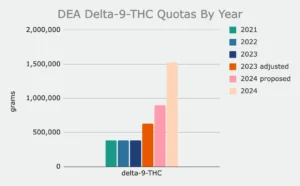
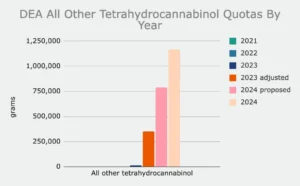
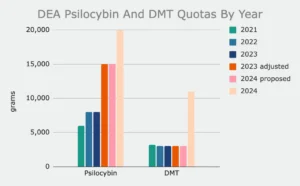
DEA is also calling for 20,000 grams of psilocybin (up from an initially proposed 15,000 grams) and 11,000 grams of DMT (up from 3,000 grams).
The production goals for marijuana and various other psychedelic compounds were kept the same as they were first proposed in November: 6,675,000 grams of marijuana, 1,000,000 grams of marijuana extract, 24,000 grams of psilocyn, 150 grams of ibogaine, 12,000 grams of MDMA, 1,200 grams of mescaline, 11,000 grams of 5-MeO-DMT and 12,000 grams of MDA.
The quotas for those substances are largely consistent with DEA’s 2023 production levels, though the agency has generally been authorizing higher amounts year-over-year as interest in the therapeutic potential of cannabis and psychedelics continues to grow.
That’s been especially true of psychedelics in more recent years, coinciding with historic local, state and federal reforms to decriminalize entheogenic plants and fungi while promoting scientific research and therapeutic access.
For example, DEA’s 2020 quota for psilocybin was 30 grams, compared to 20,000 grams in its latest proposal.
“There has been a significant increase in the use of schedule I hallucinogenic controlled substances for research and clinical trial purposes,” the agency’s earlier notice from November said. “DEA has received and subsequently approved new registration applications for schedule I researchers and new applications for registration from manufacturers to grow, synthesize, extract, and prepare dosage forms containing specific schedule I hallucinogenic substances for research and clinical trial purposes.”
In the new document, DEA also responded to comments requesting increases to psychedelic production quotas for “religious use” and expressing frustration that the agency has “disregarded their legal religious use of psychedelics as a factor when setting the production quotas of these substances.”
The commenters asked for a hearing with the DEA administrator to discuss the issue, but the agency did not directly address that request.
Instead, it pointed out that DEA has previously “held discussions with representatives of indigenous communities when requested and continued to welcome further engagement and input,” adding that production quotas are “determined in part by the individual manufacturing quota requests submitted by DEA-registered manufacturers of these substances.”
Another pair of comments recommended that the agency “include fruiting bodies containing psilocybin and psilocin and peyote buttons containing mescaline, rather than pure chemicals only” in its production quotas.
DEA responded that, because the Controlled Substances Act (CSA) has specific control over psilocybin and psilocyn, rather than the mushrooms containing those compounds, it will continue to set quotas based on those individual constituents, both synthetically and naturally derived. It said that peyote is also scheduled separately from mescaline, and the quota does not call for the production of the cacti.
Meanwhile, in 2022, DEA finally ended a longstanding monopoly on marijuana manufacturing for research purposes domestically that could presumably help meet the higher production quotas. Additionally, the agency has discussed its work to implement rules to streamline cannabis access for scientists following the enactment of a cannabis research bill in 2022.
The finalized 2024 quotas come in the context of an ongoing administrative review into the scheduling status of cannabis that DEA is actively carrying after receiving a recommendation from the U.S. Department of Health and Human Services (HHS) to move marijuana from Schedule I to Schedule III under the CSA.
 717-703-0804
717-703-0804